|
Gallium, for example, has the following electron configuration: 4 s 2 3 d 10 4 p 1. Because the electrons that occupy filled d or f subshells are seldom disturbed in a chemical reaction, we can also define an atom's valence electrons as the electrons that are not present in an atom of the preceding rare gas (ignoring filled d or f subshells). An atom's valence electrons are those electrons that can be gained or lost in a chemical reaction. The number of bonds an element can form is called valence (from the Latin valens, "to be strong"). The electrons in an outermost shell are known as valence electrons. It therefore has to gain three electrons to fill this shell. The outermost atomic orbitals for these elements are the s and p orbitals in a given shell, and it takes eight electrons in its outermost shell: 2 s 2 2 p 3. 
Sodium, for example, loses the one electron in its outermost cube at the same time that chlorine gains the electron it needs to fill its outermost cube.Īs understanding of the structure of the atom developed, it became apparent why the magic number of electrons for each of the main-group elements was eight. Lewis determined the formulas of simple ionic compounds (such as NaCI) by theorizing that atoms gain electrons if the outermost cube is more than half full, and lose electrons if the cube is less that half-full (until the cube is either full or empty). once an atom has an octet of electrons in its outermost cube, the cube becomes part of the cote of electrons about which the next cube is built.it takes eight electrons -an octet -to complete a cube.each neutral atom has one more electron in its outermost cube than the atom that precedes it in the Periodic Table.The number of electrons in the outermost cube of an atom is equal to the number of electrons lost when the atom forms positive ions.This model was based on four assumptions. All rights reserved.In 1902, while trying to find a way to explain the Periodic Table to his students, the chemist Gilbert Newton Lewis discovered that the chemistry of the main-group elements could be explained using a model in which electrons arranged around atoms are conceived as occupying the faces of concentric cubes. 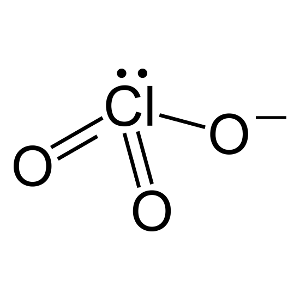
On behalf of the United States of America. Shall not be liable for any damage that may result fromįor NIST Standard Reference Data products. However, NIST makes no warranties to that effect, and NIST Uses its best efforts to deliver a high quality copy of theĭatabase and to verify that the data contained therein haveīeen selected on the basis of sound scientific judgment. The National Institute of Standards and Technology (NIST) Data from NIST Standard Reference Database 69:.Go To: Top, Gas phase ion energetics data, Referencesįree energy of reaction at standard conditionsĮnthalpy of reaction at standard conditions Mass Spectrometric Study of Thermochemical Characteristics of Perchloryl Fluoride and its Decomposition Product Chlorosyl Fluoride, Izv. Go To: Top, Gas phase ion energetics data, NotesĮxperimental and Theoretical Gas-Phase Acidities, Bond Dissociation Energies, and Heats of Formation of HClOx, x=1-4, 
Bartmess Protonation reactionsīy formula: ClO 3 - + H + = HClO 3 Quantity Secretary of Commerce on behalf of the U.S.A.ĭata compiled by: John E.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
